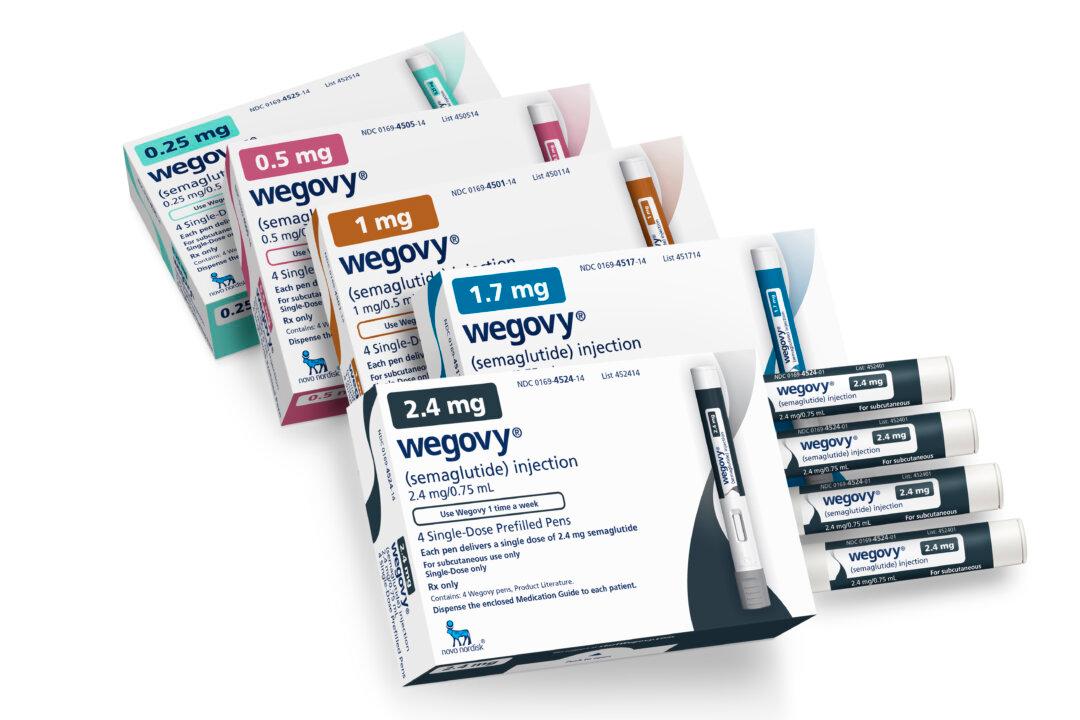
A high-profile clinical trial of the obesity drug Wegovy has raised hopes that it may also deliver significant cardiovascular benefits beyond weight loss.
The trial, funded by Wegovy’s maker Novo Nordisk, found that the use of the injected drug semaglutide led to a significant reduction in heart attacks, strokes, and cardiovascular deaths among overweight and obese patients with a history of heart disease.